Among known click chemistry, sulfur(VI) fluoride exchange (SuFEx) reaction, a new generation click chemistry, in which the S–F bond reacts readily with silyl-protected phenols, phenolates or amines. In 2019, Han Zuilhof’s group discovered that SuFEx reaction is intrinsically enantiospecific click reaction as a chiral S(VI) center reacts exclusively with inversion of configuration. The SuFEx reaction has also been successfully applied to polymer science with the efficient formation of polymers. In parallel, materials chemistry has been revolutionized by dynamic covalent chemistry (DCC), which combines the strength of covalent bonds with the power for self-healing and responsiveness that is for example displayed in supramolecular chemistry. Unlike other click chemistries, which have also shown to allow DCC to occur (like thiol-yne and Diels-Alder reactions) this has not been shown for the SuFEx reaction.
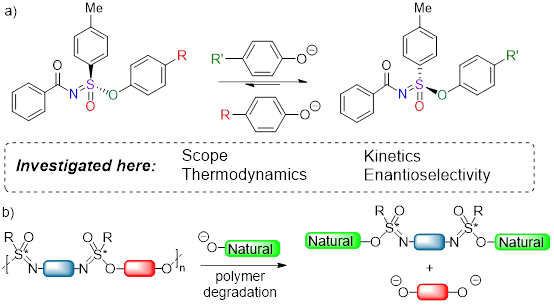
Figure 1. a) General scheme for sulfur(VI) phenolate exchange reaction, and facets under current study. b) Degradation of polysulfonimidates.
This paper describes the first case of a SuFEx-derived DCC reaction via the development of Sulfur-Phenolate Exchange (SuPhenEx) reactions, which displayed enantiospecific, wide scope, high efficiency and excellent functional group tolerance. This reaction also allows the construction of both sulfonimidate enantiomers from a single sulfonimidoyl fluoride enantiomer. Finally, SuPhenEx chemistry demonstrates that the usefulness of structurally complex, stable, yet also degradable polymer.
Relevant research was published in Angwandte Chemie International Edition, Tianjin university is the first-author institution, this work was mainly finished by Yang Chao and Akash Krishna of SPST, and the corresponding author is Prof. Han zuilhof. The work was strongly supported by all staff of the Instrumental Analytical Center, SPST, Tianjin university and Wageningen University.
The paper link: https://onlinelibrary.wiley.com/doi/10.1002/anie.202207456

