Prof. Nan Li’s team from the School of Pharmaceutical Science and Technology at Tianjin University developed an ultrasound-responsive polymeric dual-prodrug nanoassembly (PMS) co-loaded with a nitric oxide prodrug (mSNO) and polymetformin (PMet) to repair the lost fenestrae in liver sinusoids, thereby greatly improving drug delivery efficiency and opening a new avenue for liver fibrosis therapy. The related study, entitled “Ultrasound-responsive dual-prodrug nanoassembly for ‘fenestrae-restoration strategy’ in liver fibrosis therapy”, was published in Advanced Materials.
The team discovered that specifically inducing vascular endothelial cells to restore their fenestral structure, while promoting the ordered degradation of the basement membrane, can significantly increase basement membrane permeability. Inspired by this mechanism, the team proposed an innovative “fenestrae-restoration strategy”.
In liver fibrosis, excessive deposition of extracellular matrix (ECM) in the liver forms a dense “scar network” that obstructs the fenestrae on liver sinusoidal endothelial cells (LSECs), thereby impairing the normal exchange of substances between blood and hepatocytes. In addition, the basement membrane covering the outer surface of vascular endothelial cells forms a dense physical barrier, making it difficult for clinically used therapeutics to effectively penetrate from blood vessels into the deeper hepatic stellate cells (HSCs).
To address this bottleneck, Prof. Nan Li’s research group has long been dedicated to the study of liver fibrosis treatment. In a series of publications (Adv. Mater. 2024, 2406590; Adv. Funct. Mater. 2024, 2309690; Acta Pharm. Sin. B, 2025, 15(9): 4900–4916), the team has systematically investigated multiple mechanisms and potential therapeutic targets involved in liver fibrosis progression, and proposed innovative treatment strategies from different perspectives.
In this study, PMS enabled ultrasound-triggered and controllable release of nitric oxide from mSNO. This process upregulated intracellular cyclic guanosine monophosphate (cGMP), promoted reversal of LSEC capillarization, restored the fenestral structure of liver sinusoids, and enhanced endothelial permeability. Through this repair mechanism, PMet was able to penetrate the sinusoidal barrier and accumulate in fibrotic tissues, where it was subsequently taken up by HSCs. After release in lysosomes, metformin ultimately inhibited the proliferation and migration of HSCs via the AMPK-mTOR signaling pathway. These findings provide a new strategy for combination therapy in liver fibrosis.
The SPST, Tianjin University is the first corresponding institution of this paper. Prof. Nan Li is the last corresponding author, and Shutong Liu, a master’s student enrolled in 2023, is the sole first author. This work was supported by the National Natural Science Foundation of China, the Tianjin Youth Science and Technology Talent Program, and the Tianjin Natural Science Foundation. The authors also acknowledged the strong support from the Analytical Center of the SPST, Tianjin University.
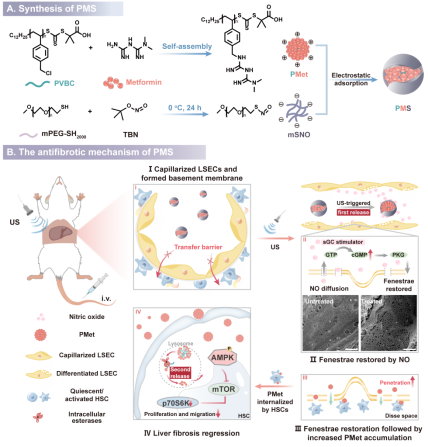
Schematic illustration of the research strategy

